 
The molecular weight can be obtained from the molecular formula, data tables, or the label on the bottle containing the chemical of interest. Molecular weight is also referred to as formula weight and, in fact, many scientists prefer to use the latter. 3 Example problem: molarity moles of solute / liters of solution 0.75 mol / 4.2 L 0.17857142 4 Write your answer. The resulting quotient will give you the number of moles per liter of solution, otherwise known as molarity. Note that V is the final or total volume of solution after the solute has been added to the solvent. Moles 0.75 mol NaCl Volume 4.2 L 3 Divide the number of moles by the number of liters. If you want to know the number of grams needed of each substance, you can multiply by the molar mass of each substance: NaOH 2.10 mol 39.997 g/mol 83.9937g Mg (OH)2 1.05 mol 58.319 g/mol 61.23495g NaCl 2.10 mol 58.442 g/mol 122. V is volume of solution in liters (L) in which the indicated mass ( m) of solute must be dissolved to make the desired molar concentration ( C).m is the mass (i.e., weight) of solute in grams (g) that must be dissolved in volume V of solution to make the desired molar concentration ( C).Using SI prefixes, the concentration may also be expressed in different fractions of the molar concentration such as mmol/L (mM), μmol/L (μM), nmol/L (nM), pmol/L (pM), etc. As mass / volume molarity molar mass, then mass / (volume molar mass) molarity. A 1 M solution is one in which exactly 1 mole of solute is dissolved in a total solution volume of exactly 1 L. Molarity is defined as the number of moles of solute dissolved per liter of solution (mol/L = M). Molarity is equal to the number of moles of a solute divided by the volume of the solution in liters. This is also referred to as molarity, which is the most common method of expressing the concentration of a solute in a solution. 1.Know the basic formula for calculating molarity. C is the molar concentration in mol/L (Molar or M). 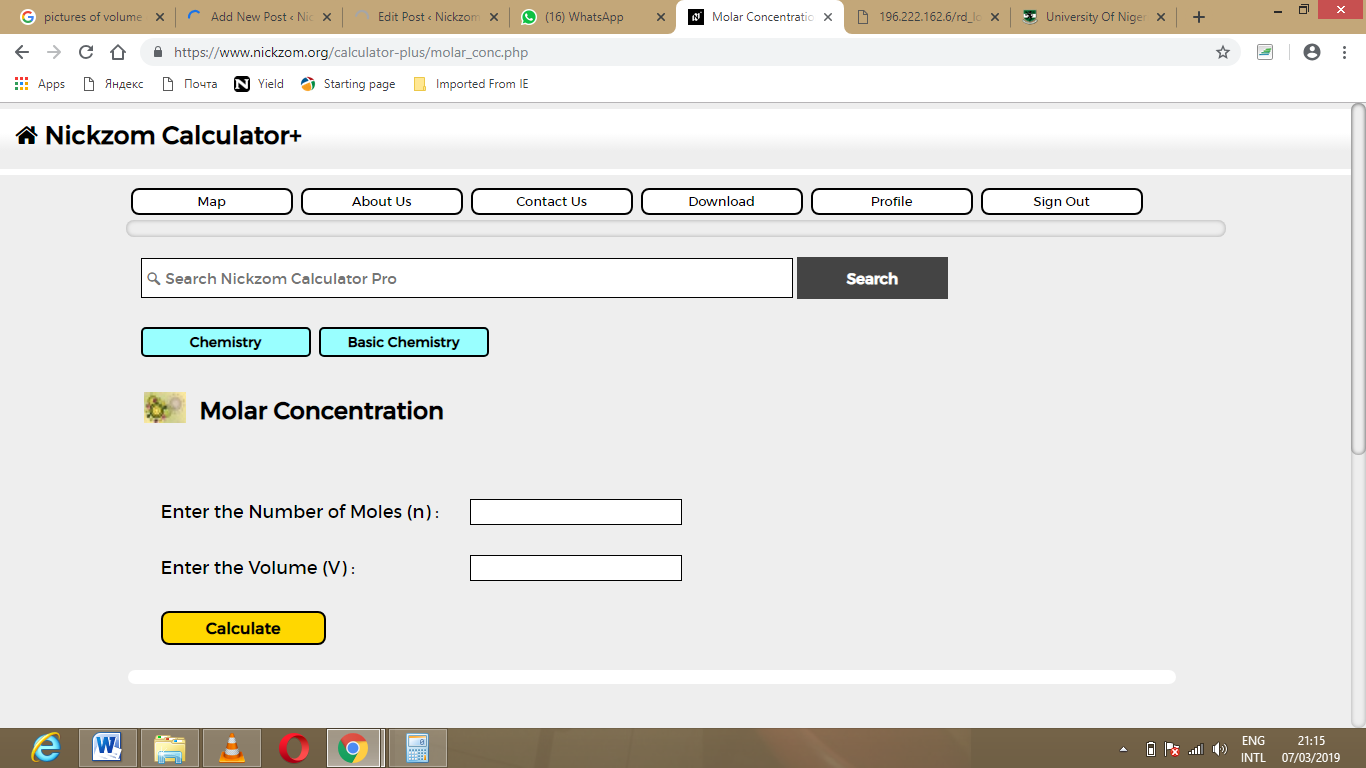
For example, a sample containing 11.2 liters (1/2 mole) of hydrogen and 11. Volume of fluorine at room temperature and pressure = 268. 1 Mol and by volume should be the same, assuming ideal gas behavior. The most commonly used unit for molarity is the number of moles per liter. Work out the volume of fluorine at room temperature and pressure. Calculate the mass or volume required to prepare a chemical solution that. Practice Question: Mary has calculated the mass of fluorine as 425g at room temperature and pressure. This leaves us with the following equation. Since moles = mass/ M r, we can substitute moles for mass/M r. We can use a different equation if we are given the mass of the gas instead of the volume. Volume = 18 dm 3 Volumes of Gases Using Mass and M r Practice Question: Calculate the volume of 0.75 mol of H 2 at room temperature and pressure. GCSE Chemistry – Volume of Gases Using Moles This means we can work out the volume of any gas at room temperature and pressure with an equation. The calculator will then instantly display the resulting volume of the ideal gas. Finally, enter the number of moles of the gas. This volume is also known as the molar gas volume, applying to all gases. Using our ideal gas volume calculator is pretty straightforward: Enter the pressure of the gas (select your preferred units first). 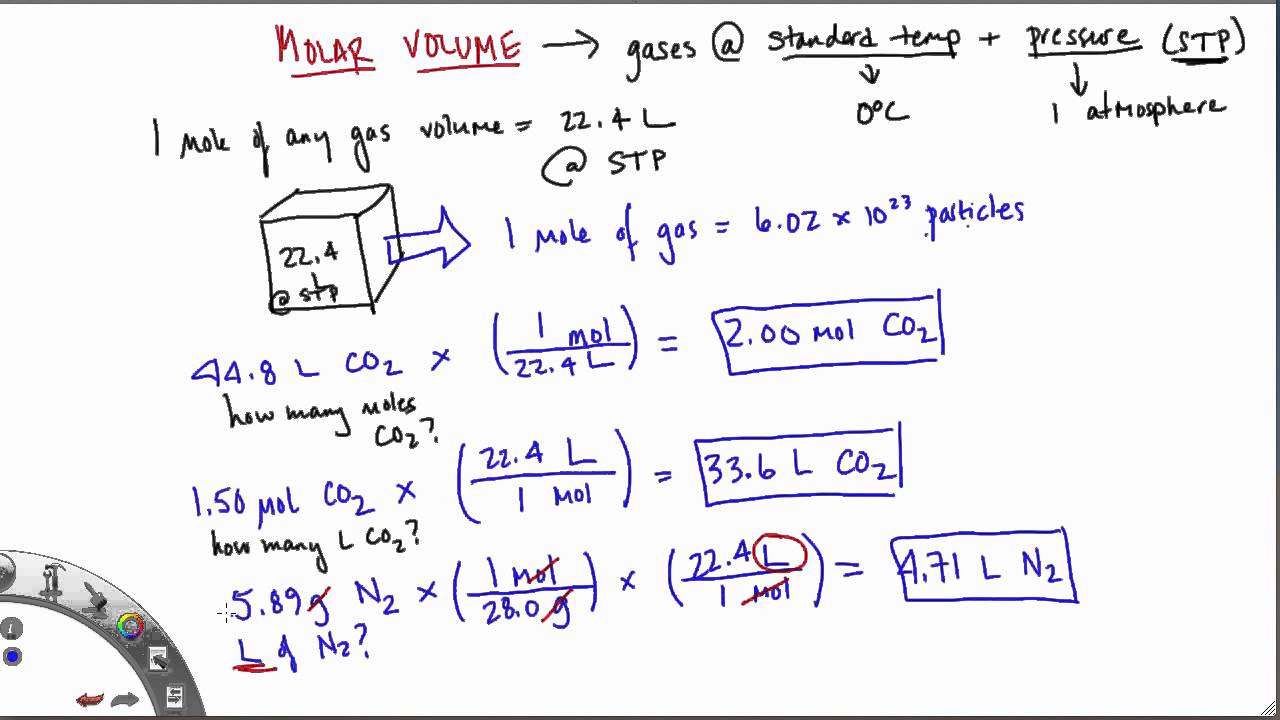
If we have one mole of gas at room temperature, then it has a volume of 24 dm 3. If gases are at room temperature (20☌) and pressure (1 atm), then they will take up 24 dm 3 of space. Volume of Gases Using Moles (GCSE Chemistry) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
